Fixing One of Healthcare’s Biggest Blind Spots With Smart Medication Technology
Modern healthcare excels at diagnosing disease and prescribing treatment, yet what happens after a prescription is written often remains invisible. Whether patients take the right medication, at the right dose, and at the right time is largely assumed rather than verified. This gap between prescription and real-world use contributes to treatment failure, safety risks, and billions of dollars in avoidable healthcare costs each year. Addressing this blind spot is the core mission of MedicaSafe.
Medication non-adherence is a systemic challenge across healthcare. Industry data cited by MedicaSafe indicate that average adherence rates hover around 72%, meaning nearly one in three doses is missed, mistimed, or misused. The consequences are especially severe in high-risk therapies such as opioid use disorder (OUD), where misuse, diversion, and inconsistent dosing significantly increase the risk of relapse and overdose. Rather than treating adherence as a behavioral failure, MedicaSafe approaches it as a design problem—one that can be solved through technology.
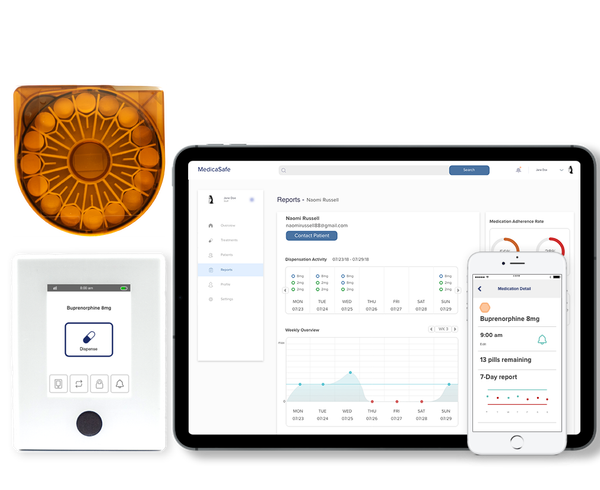
MedicaSafe’s solution integrates secure medication cartridges, controlled-access dispensing via a SmartKey system, and cloud-connected monitoring software. Medication access is restricted to the intended patient, dosing is guided and time-controlled, and every dispense event is recorded in real time. This design turns medication use from an invisible, self-reported behavior into measurable clinical data, giving providers insight into what actually happens once patients leave the clinic.
The company’s impact is most clearly demonstrated in opioid use disorder treatment programs. According to data published by MedicaSafe, patients using its system adhered to their medication regimen on 97% of days, a substantial improvement over the typical 72% adherence rate under standard care. Users also reported a satisfaction score of 6.8 out of 7, indicating strong acceptance even in stigmatized and highly sensitive treatment settings. These improvements are significant when considered at scale, particularly in the United States, where approximately 1.1 million people receive treatment for opioid use disorder each year.
While OUD has been an early focus, MedicaSafe’s platform is designed for scalability across a wide range of therapeutic areas, including mental health, chronic disease management, post-surgical recovery, and elder care. Because the system augments existing prescribing and care workflows rather than replacing them, it can be adopted without major operational disruption. Clinicians gain earlier visibility into adherence issues, allowing for timely intervention before missed doses escalate into clinical failure or emergency care.
MedicaSafe’s approach has also received institutional validation. The company has secured funding linked to the U.S. National Institutes of Health, underscoring recognition of medication non-adherence as a national public health issue. Backed by a leadership team with experience spanning healthcare, product development, and regulatory environments, MedicaSafe is positioned to navigate the complexities of scaling drug-device innovation.
By embedding safety, accountability, and data directly into medication delivery, the company reframes adherence as a solvable infrastructure problem. As healthcare systems face rising costs and increasingly complex treatments, MedicaSafe offers a compelling model for ensuring medications are not only prescribed, but taken safely, correctly, and consistently.
Learn more about the company through this LINK
Comments

E-Waste Emerges as a Major Circular Economy Challenge for Governments and Industry
Electronic waste has become one of the fastest-growing waste streams in the world, driven by rising demand for consumer electronics, shorter device lifespans, and rapid digitalization across industries. Smartphones, laptops, appliances, and connected devices are replaced at unprecedented rates, generating mounting pressure on waste systems that were never designed to
AEIR to Debut Long-Range Wireless Energy Transfer System in Zurich on December 15
Lucerne, Switzerland — AEIR announced today that it will conduct the first public demonstration of its long-range Wireless Energy Transfer (WET) system in Zurich on December 15, 2025. The debut marks a major milestone for the Swiss-based company, which has spent several years developing a technology capable of transmitting power safely
WattByWatt Uses Next-Generation Solar Materials to Power the World One Watt at a Time
As connected devices multiply and demand for clean electricity grows, the limitations of conventional solar technology are becoming more apparent. Traditional silicon panels are efficient at scale, but they are often rigid, energy-intensive to manufacture, and poorly suited for low-power or indoor applications. WattByWatt, a Canadian clean energy company, is
OceanWell Taps Deep Ocean to Redefine Sustainable Desalination
Water scarcity is rapidly becoming one of the most pressing global challenges, driven by climate change, population growth, and declining freshwater reserves. While desalination has long been seen as part of the solution, conventional systems remain energy-intensive, land-heavy, and environmentally controversial. OceanWell is advancing a new approach that rethinks how
Revolutionizing Water Management: How Technology is Tackling Water Waste in Buildings.
Shayp client ING went live in 2022 and has already saved over 3 million litres per year. In a couple of months, they were able to avoid 473 kg of carbon emissions by just monitoring their water and taking action efficiently.
